Some lotions remain as a partitioned mix of two oil-based and water-based halves. Some examples are emulsifying wax, which is wax-treated with a detergent-like solution, and ceteareth, a substance that is a combination of saturated fatty alcohols. Additional substances called emulsifiers are often used to stabilize the emulsion - this is commonly the case in cosmetic products.
Emulsions are mixtures of several substances that do not mix to form a homogeneous solution, such as oil and water. All three are emulsions of water and oils, and higher viscosity is a result of higher oil content. Lotions, creams, and ointments, whether medicated or not, are distinguished by their viscosity. Controlling it saves lives, helps generate lucrative profits to the manufacturers of cosmetics, and makes cooking easier.
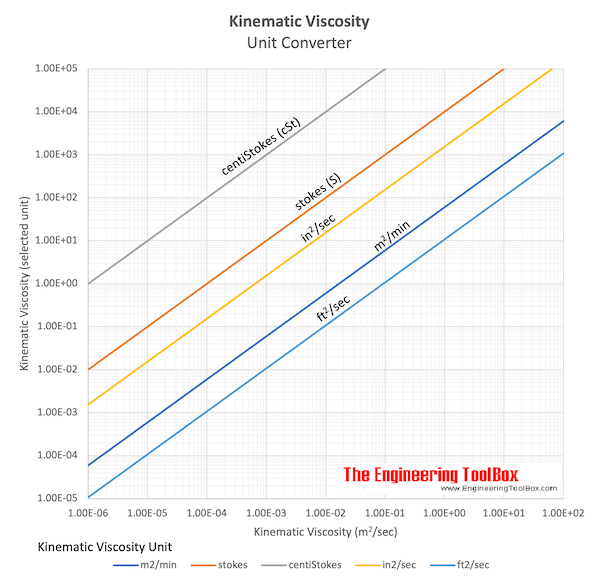
Viscosity conversion how to#
Understanding viscosity and how to control it is very important in a range of industries. When you stir the cream with a spoon, its viscosity is low, but when you beat it with a mixer, its viscosity increases gradually, and eventually it gets so viscous that it becomes stiff enough to keep its shape. Cream is a good example of a non-Newtonian fluid because it is not very viscous under normal conditions or if some small amount of force is applied to it, but becomes very viscous if the force is intensified. Non-Newtonian fluids become either more viscous or less viscous depending on how the force is applied to them. Newtonian fluids do not change the degree of viscosity based on the force that acts upon them. It is important to note the difference between Newtonian and non-Newtonian fluids when talking about viscosity. Water is a Newtonian fluid Newtonian and Non-Newtonian Fluids Olive oil is not viscous at room temperature, but it becomes viscous when refrigerated. For example, warm honey is easy to stir but is more viscous when colder, and so are some oils. The viscosity of many substances is temperature-dependent, therefore it is important to specify temperature when providing viscosity information. High viscosity refers to high resistance to shear stress. Solids that undergo small continuous stress, deform similar to liquids and could be described in terms of viscosity, but this property is most often used in relation to liquids and gases. The property of the jam to follow your hand is viscosity - the jam does not flow, even though you push it, but follows your hand instead. The jam will follow your hand and the cube will be distorted. If you put this jam cube onto a plate, then touch the top of the cube with your hand, and pull your hand parallel to the surface of the cube in any direction - you will be acting on the jam cube with a force that causes shear stress. For example, imagine a substance so viscous, that it can retain its shape, say a cube, for several minutes almost without changing - like a very viscous fruit jam. Viscosity is often talked about in the context of shear stress, where shear stress is defined as stress that results from a force that acts parallel to the cross-section of the object.
Dynamic or absolute viscosity measures the resistance inside of the fluid and is independent of the density of this fluid.
Viscosity is a property of a fluid to resist the force that pushes the fluid to flow. Pouring viscous cream into a cup of non-viscous coffee
